Closing the Carbon Cycle With Green Propane Production
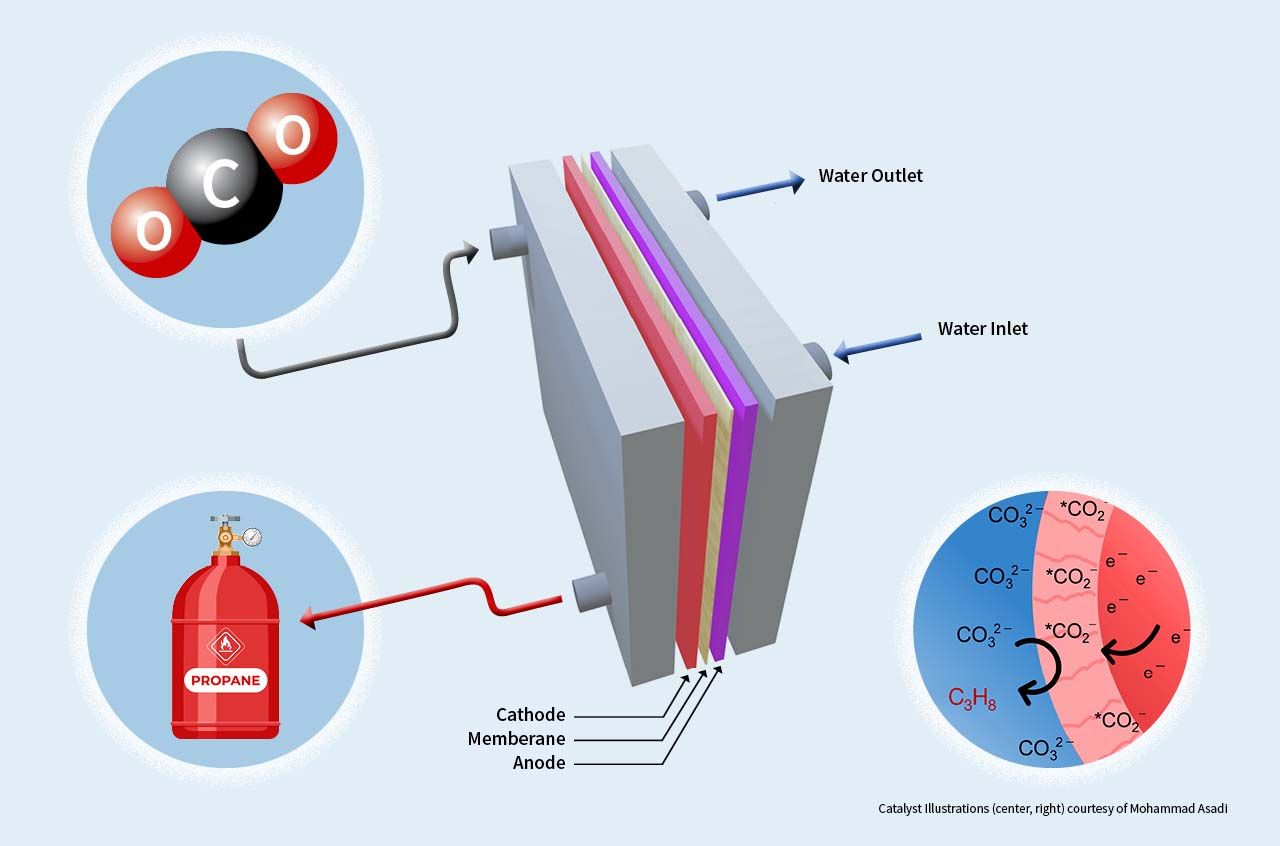
Illinois Institute of Technology Assistant Professor of Chemical Engineering Mohammad Asadi has published a paper in Nature Energy that describes an electrolyzer device that can convert carbon dioxide into propane through a method that is economically feasible at a large scale.
The United States has set a target of net-zero greenhouse gas emissions by 2050. Electric power and industry sectors account for around half of U.S. carbon dioxide emissions, so advances to reduce these emissions are an urgent goal.
“Making renewable chemical manufacturing is really important,” says Asadi. “It’s the best way to close the carbon cycle without losing the chemicals we currently use daily.”
The key to Asadi’s electrolyzer is a one-of-a-kind catalytic system that uses inexpensive, readily available materials to produce tri-carbon molecules, a core component of many fuels such as propane, which is used in a range of applications including home heating and aviation fuel.
The study uses experimental and computational methods to provide an in-depth understanding of how the catalyst works at a fundamental level, including identifying which factors play key roles in reaction activity, selectivity, and stability.
Asadi says one of the reasons this technology is more commercially viable than other works is that the group used a flow electrolyzer, which allows for the continuous production of propane, compared to other technologies that require batch processing.
“Designing and engineering this laboratory-scale flow electrolyzer prototype has demonstrated Illinois Tech’s commitment to creating innovative technologies. Optimizing and scaling up this prototype will be an important step toward producing a sustainable, economically viable, and energy-efficient carbon capture and utilization process,” says Advanced Research Projects Agency-Energy Program Director Jack Lewnard.
Asadi has previously used a version of this catalyst to produce ethanol by extracting carbon dioxide from industrial waste gas.
Having proven the technology’s success, Asadi has partnered with SHV Energy, a leading global propane distributor, to scale up the system and make it more widely available.
“This is an exciting development which opens up a new e-fuel pathway to on-purpose propane production for the benefit of global users of this essential fuel,” says Keith Simons, head of research and development for sustainable fuels at SHV Energy.
Illinois Tech Duchossois Leadership Professor and Professor of Physics Carlo Segre, University of Pennsylvania Professor of Materials Science and Engineering Andrew Rappe, and University of Illinois Chicago Professor Reza Shahbazian-Yassar contributed to this work. Mohammadreza Esmaeilirad (Ph.D. CHE ’22) was a lead author on the paper.
Disclaimer: “Research reported in this publication was supported by the National Science Foundation under Award Number 2135173, the Advanced Research Projects Agency-Energy under Award Number DE-AR0001581, and SHV Energy. This content is solely the responsibility of the authors and does not necessarily represent the official views of the National Science Foundation, the Advanced Research Projects Agency-Energy, or SHV Energy.”
Mohammad Asadi, “Imidazolium Functionalized Transition Metal Phosphide Catalysts for Electrochemical Carbon Dioxide Conversion to Ethanol,” National Science Foundation; Award Number 2135173
Mohammad Asadi, “Direct Conversion of Flue Gas to Value-Added Chemicals Using a Carbon-Neutral Process,” Advanced Research Projects Agency-Energy; Award Number DE-AR0001581
Photo: Illustration of electrolyzer, which uses a novel catalyst to convert carbon dioxide into propane.




